Biotech
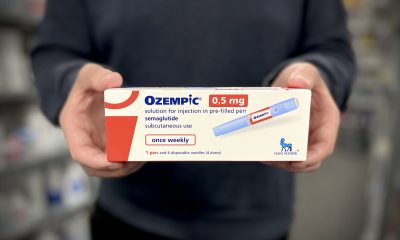
Eli Lilly Warns of Safety Risks in Compounded Tirzepatide Mixed with Vitamin B12
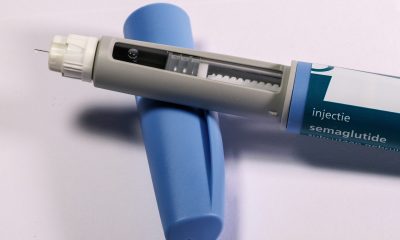
Eli Lilly warns that compounded tirzepatide mixed with vitamin B12 may form harmful impurities with unknown health effects. These untested combinations lack safety data and regulatory oversight. Patients using such products face potential risks, including contamination and toxicity. Lilly urges consultation with doctors and supports stronger regulatory action against illegally compounded tirzepatide products to protect public safety.

Eli Lilly has issued a public warning regarding potential safety risks linked to compounded tirzepatide products mixed with vitamin B12.
As part of its ongoing commitment to patient safety, the biotech company conducted tests on compounded products marketed in the United States that combine tirzepatide with various forms of vitamin B12, including methylcobalamin, hydroxocobalamin, and cyanocobalamin. These tests identified significant levels of an impurity formed through a chemical reaction between tirzepatide and vitamin B12.
Unknown impurities and untested combinations raise serious concerns for patient safety, Eli Lilly says
This impurity raises serious concerns because its effects on humans remain entirely unknown. There is no available data on its short- or long-term impact, potential toxicity, or possible immunological reactions.
Additionally, it is unclear how this impurity interacts with GLP-1 and GIP receptors, or how it is absorbed, distributed, metabolized, and eliminated by the body. Tirzepatide has never been clinically studied in combination with vitamin B12, leaving a significant gap in safety knowledge. Compounding manufacturers are also not required to monitor or report adverse events, further increasing uncertainty around these products.
Eli Lilly cautions that individuals receiving tirzepatide-B12 combinations from compounding pharmacies, telemedicine providers, medspas, or other sources may be exposed to potentially dangerous products with unknown risks. The company has informed the U.S. Food and Drug Administration (FDA) of its findings and advises patients currently using such products to consult their healthcare providers to discuss safer, approved treatment alternatives.
The company has consistently voiced concerns about the safety of mass-produced tirzepatide imitations, including those marketed as alternatives to approved medications. Lilly supports the FDA’s recent announcement of plans to take stronger action against the widespread distribution of illegally compounded anti-obesity drugs. The FDA has repeatedly warned that compounded drugs carry greater risks than approved medications, as they are not subject to the same rigorous evaluation for safety, effectiveness, and quality.
Despite regulatory and legal efforts to halt the mass production of tirzepatide, some manufacturers continue to distribute these products under the label of “customized” treatments. In many cases, these so-called customized formulations simply involve adding untested substances, such as vitamin B12, to tirzepatide. According to Eli Lilly, these additions are not genuinely tailored to individual patients but are instead broadly applied in an attempt to bypass regulatory restrictions. Testing indicates that such combinations may introduce even greater risks than previously understood.
The issue extends beyond vitamin B12. Other additives, including glycine, pyridoxine, niacinamide, and carnitine, are also being mixed with tirzepatide without proper evaluation. These substances have no proven clinical benefit when combined with tirzepatide and may create new, untested drug combinations with unpredictable effects. Eli Lilly has also identified additional safety concerns in compounded tirzepatide products, such as bacterial contamination, elevated endotoxin levels, and other impurities not found in FDA-approved medications.
Eli Lilly emphasizes that the widespread distribution of these unassessed compounded drugs represents a significant and unacceptable risk to patients. The company urges regulators to continue enforcing existing laws and calls for the recall of compounded tirzepatide products containing untested additives like vitamin B12.
__
(Featured image by Kobe Tang via Unsplash)
DISCLAIMER: This article was written by a third party contributor and does not reflect the opinion of Born2Invest, its management, staff or its associates. Please review our disclaimer for more information.
This article may include forward-looking statements. These forward-looking statements generally are identified by the words “believe,” “project,” “estimate,” “become,” “plan,” “will,” and similar expressions. These forward-looking statements involve known and unknown risks as well as uncertainties, including those discussed in the following cautionary statements and elsewhere in this article and on this site. Although the Company may believe that its expectations are based on reasonable assumptions, the actual results that the Company may achieve may differ materially from any forward-looking statements, which reflect the opinions of the management of the Company only as of the date hereof. Additionally, please make sure to read these important disclosures.
First published in GACETA MEDICA. A third-party contributor translated and adapted the article from the original. In case of discrepancy, the original will prevail.
Although we made reasonable efforts to provide accurate translations, some parts may be incorrect. Born2Invest assumes no responsibility for errors, omissions or ambiguities in the translations provided on this website. Any person or entity relying on translated content does so at their own risk. Born2Invest is not responsible for losses caused by such reliance on the accuracy or reliability of translated information. If you wish to report an error or inaccuracy in the translation, we encourage you to contact us.

-

 Crypto2 weeks ago
Crypto2 weeks agoCrypto Markets Volatile as Bitcoin Hovers, Solana Eyes AI Integration
-

 Cannabis17 hours ago
Cannabis17 hours agoGermany’s Cannabis Reform Shows Limited Risks but Calls for Regulatory Adjustments
-
 Biotech1 week ago
Biotech1 week agoAdvances in LDL Cholesterol Management for Familial Hypercholesterolemia: From Statins to Innovative PCSK9 and CETP Inhibitors
-

 Business5 days ago
Business5 days agoTopRanked.io Weekly Affiliate Digest: What’s Hot in Affiliate Marketing [Bovada Affiliate Program]