Biotech
Novo Nordisk Introduces Norditropin Nordiflex, a Device for Growth Hormone Administration
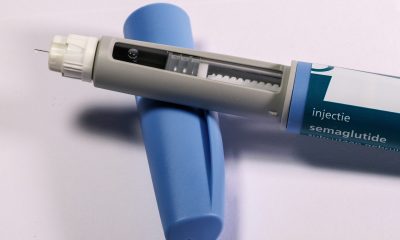
Currently, in Spain, it is estimated that around 3% of the population under 15 years of age has a short stature that may be related to a growth disorder. In total, some 200,000 children. Novo Nordisk introduces Norditropin Nordiflex, a new device for growth hormone treatment. The incorporation of new functionalities brings with it an improvement in the patient’s quality of life.

Novo Nordisk introduces Norditropin Nordiflex, a new device for growth hormone treatment. Thus, this solution for injection in a pre-filled pen is indicated for the treatment with somatropin (growth hormone) of growth retardation in children who meet the indication for growth hormone deficiency (GHD), children born small for gestational age (SGA), Turner syndrome (TS), chronic renal failure (CRF), Noonan syndrome (NS) and adults with GDH either with onset in childhood or in adulthood.
Read more on the subject and find the most important business headlines with the Born2Invest mobile app.
Novo Nordisk introduces Norditropin Nordifle, a new device to administer growth hormone
Currently, in Spain, it is estimated that around 3% of the population under 15 years of age has a short stature that may be related to a growth disorder. In total, some 200,000 children. “The start of treatment at an early age is always difficult, both for the child and for the family. The most shocking thing for them is the fact of having to prick themselves, so making it as easy as possible for them to administer the treatment is a benefit that in the long term will lead to better results,” said Dr. Rafael Espino, head of pediatric endocrinology at the Virgen de Valme University Hospital and head of the Pediatrics Department at the Quirónsalud Infanta Luisa Hospital in Seville.
Elena Garrido, a nurse at the Vall de Hebrón University Hospital in Barcelona, explained that “this new injectable solution provides simplicity and convenience thanks to a soft push button. In addition, this new device can be coupled with PenMate, which eliminates the anguish of the needle stick, since it contains a needle concealer with an automatic self-injection method. So it’s more convenient and easier.”
Children, the main target group for growth hormone treatment, are the main beneficiaries
Another important aspect is the preparation of the product to be administered. According to Dr. Espino, “with Norditropin Nordiflex we are taking an important step forward since the device comes prepared with the growth hormone inside. In other words, it is ready for use, with no assembly required. So, it is very easy to learn to use. And then there is the speed. With it, it is no longer necessary to count the “clicks” because the dose is marked in mg. At the same time, it is very precise and flexible because the dose increments are adjusted to the needs of each patient, avoiding overdosing.”
These advantages are very important considering that the starting age for treatment is between 4-5 years of age. In this sense, Elena Garrido pointed out that “at this age, the more facilities we provide for administering the treatment, both to them and to their families, the better the adherence will be and, consequently, the greater the benefits”. In any case, the causes of growth retardation are multiple, and, depending on them, the age of onset of growth disorders can vary.
Autonomy and greater adherence
The incorporation of all these new functionalities brings with it an improvement in the patient’s quality of life. “It is a device that is also very easy to transport. Moreover, it can be kept for 21 days at room temperature up to 25°C. In this way, it not only favors the child’s autonomy but also provides greater peace of mind for the parents or relatives who are caring for the child,” said Elena Garrido.
Dr. Espino added that “it is also very important to start treatment early and to adhere to it. If we manage to control the treatment well and not to skip any doses, then, obviously, the results and prognosis will be better in less time. All these improvements in the patient experience have an impact on better adherence.
According to Francisco Pajuelo, medical director of Novo Nordisk Spain, “Norditropin Nordiflex is another example of Novo Nordisk’s commitment to constant innovation that contributes to solving patient needs in a faster and simpler way. An innovation that is also part of the company’s commitment to improving the lives of people with growth disorders through therapeutic options that are the result of listening to and learning from them and the professionals who care for them”.
The new device provides greater flexibility and accuracy by allowing adjustment of the optimal dosage range prescribed by the physician. Height below the percentile for the child’s age and genetic prospects is the main warning sign of a possible growth disorder. As Dr. Espino points out, “short stature is the main reason for consultation in pediatric endocrinology. Of the various indications for growth hormone treatment, the most common is GHD and PEG”.
Specifically, continues this expert, “GHD is for children with idiopathic growth hormone deficiency, but we are unable to identify the exact cause. In other words, they are patients who have been very well studied and meet the criteria required for treatment. This is the case for almost two out of three children currently receiving treatment. The other third are, in general, those who are SGA, so they should be identified at birth”.
Both experts agreed that “in general, most children who are treated with growth hormone respond favorably to treatment as long as it is carried out correctly. So, all innovations that help this are welcome.
__
(Featured image by Bessi via Pixabay)
DISCLAIMER: This article was written by a third party contributor and does not reflect the opinion of Born2Invest, its management, staff or its associates. Please review our disclaimer for more information.
This article may include forward-looking statements. These forward-looking statements generally are identified by the words “believe,” “project,” “estimate,” “become,” “plan,” “will,” and similar expressions. These forward-looking statements involve known and unknown risks as well as uncertainties, including those discussed in the following cautionary statements and elsewhere in this article and on this site. Although the Company may believe that its expectations are based on reasonable assumptions, the actual results that the Company may achieve may differ materially from any forward-looking statements, which reflect the opinions of the management of the Company only as of the date hereof. Additionally, please make sure to read these important disclosures.
First published in iSanidad, a third-party contributor translated and adapted the article from the original. In case of discrepancy, the original will prevail.
Although we made reasonable efforts to provide accurate translations, some parts may be incorrect. Born2Invest assumes no responsibility for errors, omissions or ambiguities in the translations provided on this website. Any person or entity relying on translated content does so at their own risk. Born2Invest is not responsible for losses caused by such reliance on the accuracy or reliability of translated information. If you wish to report an error or inaccuracy in the translation, we encourage you to contact us.

-

 Business7 days ago
Business7 days agoThe TopRanked.io Travel Affiliates Guide [+ Canadian Travel Opportunity]
-

 Biotech2 weeks ago
Biotech2 weeks agoAbivax Emerges as Prime Takeover Target in Booming IBD Market
-

 Crowdfunding2 days ago
Crowdfunding2 days agoLa Vía Campesina Launches Global Solidarity Campaign for Cuba Amid Blockade Criticism
-

 Impact Investing1 week ago
Impact Investing1 week agoSustainability in the Food Sector Becomes a Competitive Requirement