Biotech
Pharma Mar’s Sylentis will conduct a trial in more than 30 hospitals in the U.S.
Sylentis is preparing for a new trial. The study has been approved by the FDA in 30 days and will be part of the marketing authorization application. Tivanisiran represents a breakthrough in the development of innovative drugs in different therapeutic areas through gene silencing technology based on RNA interference (RNAi). The trial will take place in 30 US hospitals.
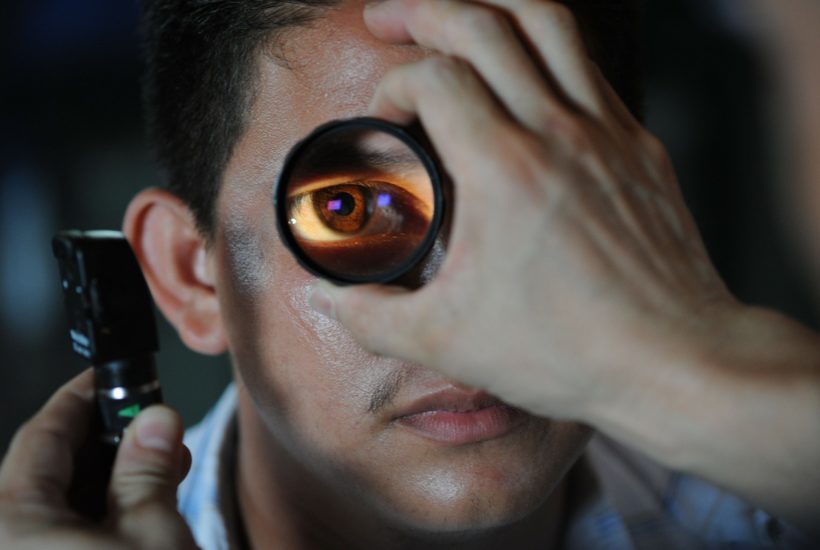
Pharma Mar’s Sylentis will conduct a trial in more than 30 U.S. hospitals. The PharmaMar group company announced Thursday, March 4th, that the U.S. Food and Drug Administration (FDA) has authorized the start of a Phase III clinical trial with the ophthalmic solution (eye drops) tivanisiran for the treatment of dry eye disease associated with Sjögren’s syndrome.
The study design has been approved by the FDA in 30 days and will be part of the marketing authorization application, which will include another Phase III trial. This study will involve more than 30 hospitals in the United States and 200 patients. It is a randomized, double-masked, placebo-controlled study, whose primary and secondary objectives are, respectively, to evaluate the efficacy (signs and symptoms) and safety of tivanisiran in patients with dry eye disease associated with Sjögren’s Syndrome. This Phase III trial will not be conducted in Spain.
If you want to find more details about the new trial conducted by Sylentis in 30 US hospitals and to find other important breakthroughs in the biotech sector, download for free the Born2Invest mobile app. Our companion app is available for both Android and iOS devices and keeps its readers up to date with the most important business news of the day.
Sylentis receives FDA green light for a trial of its ophthalmic solution for dry eye disease
Tivanisiran represents a breakthrough in the development of innovative drugs in different therapeutic areas through gene silencing technology based on RNA interference (RNAi). It is a drug administered in drops to control the inflammation and pain characteristic of this pathology.
Both the design of the protocol for this trial and its authorization by the regulatory authorities are based on the scientific evidence of safety and efficacy obtained in the phase III Helix trial. In this study, a significant improvement in signs and symptoms was observed in the subgroup of patients with Sjögren’s syndrome, which is a more severe form of dry eye disease.
In the words of Ana Isabel Jiménez, head of research and development (R&D) at Sylentis, “the start of this trial in the United States represents an important step towards advancing our goal of offering innovative therapies to patients with ocular surface diseases.”
Pharma Mar closed 2020 with revenues of $165 million (€137 million)
Pharma Mar achieved its best-ever results in 2020, with revenues of $165 million (€137 million), compared with a loss of $10.9 million (€9.1 million) in 2019. The group tripled its revenues in 2020 to $325 million (€270 million), compared with the $103 million (€85.8 million) posted in 2019, representing growth of 215%.
Total oncology sales were $120.4 million (€100 million), up 38% from a year earlier. Pharma Mar recorded its first revenues from sales of lurbinectedin on compassionate use in certain European countries in 2020. This item recorded total revenues of $25.9 million (€21.5 million). In addition, royalty revenues jumped by 405% to $18.8 million (€15.6 million).
__
(Featured image by 12019 via Pixabay)
DISCLAIMER: This article was written by a third party contributor and does not reflect the opinion of Born2Invest, its management, staff or its associates. Please review our disclaimer for more information.
This article may include forward-looking statements. These forward-looking statements generally are identified by the words “believe,” “project,” “estimate,” “become,” “plan,” “will,” and similar expressions. These forward-looking statements involve known and unknown risks as well as uncertainties, including those discussed in the following cautionary statements and elsewhere in this article and on this site. Although the Company may believe that its expectations are based on reasonable assumptions, the actual results that the Company may achieve may differ materially from any forward-looking statements, which reflect the opinions of the management of the Company only as of the date hereof. Additionally, please make sure to read these important disclosures.
First published in PlantaDoce, a third-party contributor translated and adapted the article from the original. In case of discrepancy, the original will prevail.
Although we made reasonable efforts to provide accurate translations, some parts may be incorrect. Born2Invest assumes no responsibility for errors, omissions or ambiguities in the translations provided on this website. Any person or entity relying on translated content does so at their own risk. Born2Invest is not responsible for losses caused by such reliance on the accuracy or reliability of translated information. If you wish to report an error or inaccuracy in the translation, we encourage you to contact us.

-

 Africa2 weeks ago
Africa2 weeks agoMorocco’s Mining Ambitions: From Isolated Hubs to Integrated Industrial Power
-

 Fintech12 hours ago
Fintech12 hours agoPayPal Settles US Discrimination Probe and Revises Small Business Program
-

 Africa1 week ago
Africa1 week agoMorocco’s Budget Deficit Widens to 15.5 Billion Dirhams by April 2026
-

 Markets3 days ago
Markets3 days agoCocoa Markets Face Rising Supply and Weak Demand Pressure