Biotech
Ability Pharma Receives 2 Million From the Government for Its Cancer Treatment
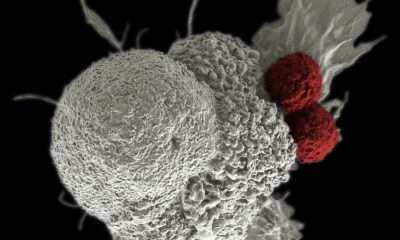
Ability Pharma is working on a drug capable of inducing cancer cell death through autophagy. ABTL0812 has demonstrated clinical efficacy in a Phase II clinical trial in patients with endometrial cancer or non-small cell lung cancer, as first-line treatment in combination with chemotherapy and as maintenance treatment after chemotherapy cycles.

Financial backing for Abilty Pharma. The Spanish biopharmaceutical company has received €2 million from the government to advance its anticancer treatment, according to Carles Domènech, CEO of Ability Pharma.
The Catalan company has received the amount as non-dilutive funding from the Ministry of Science and Innovation to accelerate the regulatory pathway for its anti-cancer autophagy marker ABTL0812, Domènech added. The drug has been shown to be able to induce cancer cell death through autophagy (self-digestion).
ABTL0812 has demonstrated clinical efficacy in a Phase II clinical trial in patients with endometrial cancer or non-small cell lung cancer, as first-line treatment in combination with chemotherapy and as maintenance treatment after chemotherapy cycles.
The drug has also received orphan drug designation (ODD) for pancreatic cancer, biliary cancer, and pediatric neuroblastoma from the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Read more about Ability Pharma and find other important market updates with our companion app. Born2Invest mobile application is bringing the latest biotech and business news from trusted sources to a single screen so you can stay on top of the market. The application is aggregating the most important and breaking news from relevant websites, the list is always revised and updated with new resources.
Ability Pharma is working on a drug capable of inducing cancer cell death through autophagy
In preclinical cancer models, ABTL0812 is effective as a single agent with a safety profile across a broad spectrum of cancer types, including lung, endometrial and pancreatic cancer, neuroblastoma, and glioma.
In 2021, the company announced the inclusion of the first patients in a Phase 2b clinical trial with ABTL0812 and another drug, Folfirinox, as first-line therapy in advanced pancreatic cancer simultaneously in the United States and Europe. The study started simultaneously at the University of Cincinnati Medical Center and Vall d’Hebron Institut d’Oncologia, with Davendra Sohal and Teresa Macarulla as principal investigators.
The trial was funded with €5 million from the European Commission’s EIC Accelerator Program under Horizon 2020 and $2 million from the NIH-R01 grant managed by the FDA in its Orphan Disease Program.
Ability Pharma is a biopharmaceutical company founded in 2009, based in Cerdanyola del Vallès (Barcelona), and focused on the development of innovative oral anticancer compounds that produce selective autophagy-mediated cytotoxicity in cancer cells.
The ABTL0812 molecule is the most advanced active and has already been positively tested in Phase 2 clinical trials as first-line therapy in patients with endometrial or squamous lung cancer in Europe.
Ability Pharma’s shareholders include venture capital funds Inveready, Fitalent (Everis Group), SciClone Pharmaceuticals and its founders. The company is also supported by Acció, EIC Accelerator, Enisa and Mineco.
__
(Featured image by ernestoeslava via Pixabay)
DISCLAIMER: This article was written by a third party contributor and does not reflect the opinion of Born2Invest, its management, staff or its associates. Please review our disclaimer for more information.
This article may include forward-looking statements. These forward-looking statements generally are identified by the words “believe,” “project,” “estimate,” “become,” “plan,” “will,” and similar expressions. These forward-looking statements involve known and unknown risks as well as uncertainties, including those discussed in the following cautionary statements and elsewhere in this article and on this site. Although the Company may believe that its expectations are based on reasonable assumptions, the actual results that the Company may achieve may differ materially from any forward-looking statements, which reflect the opinions of the management of the Company only as of the date hereof. Additionally, please make sure to read these important disclosures.
First published in PlantaDoce, a third-party contributor translated and adapted the articles from the originals. In case of discrepancy, the original will prevail.
Although we made reasonable efforts to provide accurate translations, some parts may be incorrect. Born2Invest assumes no responsibility for errors, omissions or ambiguities in the translations provided on this website. Any person or entity relying on translated content does so at their own risk. Born2Invest is not responsible for losses caused by such reliance on the accuracy or reliability of translated information. If you wish to report an error or inaccuracy in the translation, we encourage you to contact us.

-

 Crypto2 weeks ago
Crypto2 weeks agoCrypto Holds Steady as Tether Backs DRIFT and Schwab Eyes Market Entry
-

 Impact Investing1 day ago
Impact Investing1 day agoEU Parliament Advances ETS2 Plan to Stabilize Carbon Market for Transport and Buildings
-

 Biotech1 week ago
Biotech1 week agoAI and Innovation Transform Clinical Practice in Spanish Hospitals, Despite Ongoing Challenges
-

 Crypto4 days ago
Crypto4 days agoGermany’s Crypto Tax Rules and Bitcoin Outlook Shape Investor Strategy